* ADC (Antibody-Drug Conjugate) background
The “Smart Bomb” or “Guided Missile” for cancer.
For those new to the world of biotechnology, ADC (Antibody-Drug Conjugate) can be understood through a very simple and powerful analogy: the “Smart Bomb” or “Guided Missile” for cancer.
Traditionally, chemotherapy was like a “grenade”—it killed cancer cells, but it also damaged many healthy cells, leading to severe side effects like hair loss and nausea. ADC was designed to solve this problem by combining the precision of a “biological GPS” with the power of a “lethal warhead.”
The 3 Core Components of an ADC
To understand an ADC, imagine a package delivered by a drone. It consists of three essential parts:
- The Antibody (The GPS / Homing Device): This is a protein designed to find a specific “address” (a marker called an antigen) that is only found on the surface of cancer cells. It ignores healthy cells and heads straight for the tumor.
- The Payload (The Warhead / Explosive): This is a highly potent anti-cancer drug. These drugs are often so powerful that they cannot be injected into the bloodstream alone because they would be too toxic. By “hiding” them on the antibody, we can transport them safely.
- The Linker (The Smart Switch): This is the “glue” that connects the antibody and the drug. Its job is to stay rock-solid in the blood but snap open as soon as the package is inside the cancer cell. This is where companies like LigaChem Bio specialize—creating a linker that never leaks early.
How an ADC Works (The 3-Step Mission)
- Targeting: Once injected, the ADC travels through the bloodstream until the Antibody finds the cancer cell and locks onto its surface.
- Internalization (The Trojan Horse): The cancer cell “swallows” the ADC, thinking it is a nutrient. This is exactly like the Greek myth of the Trojan Horse—the “soldiers” (drugs) enter the “city” (cancer cell) hidden inside.
- Release and Destruction: Once inside, the Linker detects the specific environment of the cancer cell and releases the Payload. The drug then destroys the cancer cell from the inside out, leaving the neighboring healthy cells untouched.
Why Is Everyone Talking About ADC?
- Safety: Because the drug is only released inside the target, patients experience significantly fewer side effects compared to traditional chemotherapy.
- Potency: It allows doctors to use much stronger drugs than ever before, which can kill even the most stubborn, resistant cancer cells.
- Versatility: By simply changing the “Antibody,” scientists can create different ADCs for breast cancer, lung cancer, stomach cancer, and more.
Review of LigaChem Bio’s ADC Platform
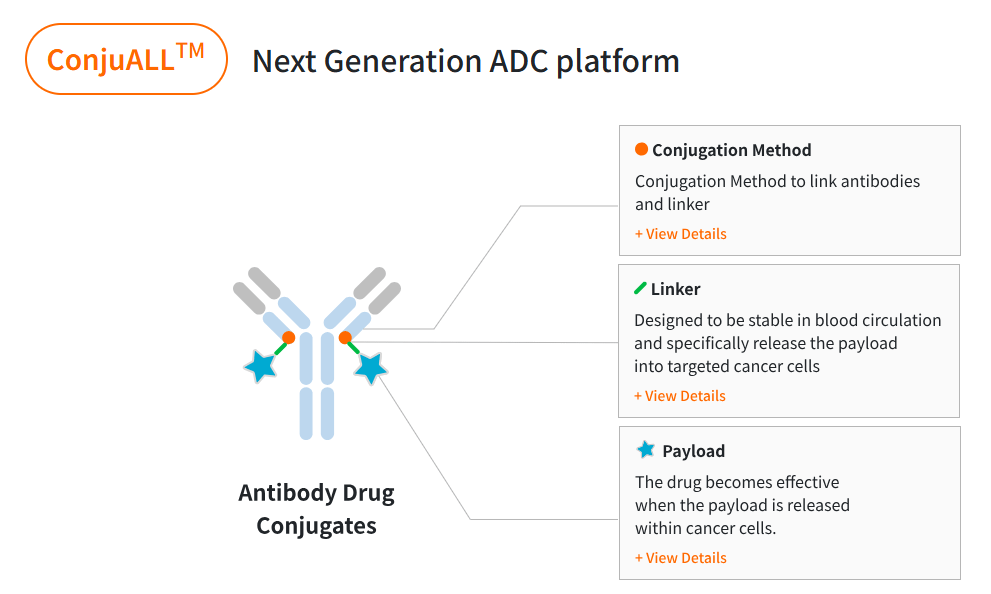
LigaChem Bio (LCB) has redefined the architecture of Antibody-Drug Conjugates (ADCs) through its proprietary ConjuAll™ platform. While traditional ADCs often suffer from “systemic leakage” and “heterogeneity,” LCB’s technology focuses on three pillars of engineering excellence: Site-Specific Conjugation, Linker Stability, and Next-Generation Payloads.
1. Site-Specific Conjugation: Achieving Molecular Homogeneity
Conventional ADCs are often produced via random conjugation, resulting in a mixture of antibodies with varying numbers of drugs attached (a wide distribution of DAR, Drug-to-Antibody Ratio). This heterogeneity leads to inconsistent efficacy and unpredictable safety profiles.
- The LCB Advantage: LCB utilizes a unique enzymatic method to attach payloads only to specific, engineered sites on the antibody.
- DAR Control: This allows for the production of ADCs with a precise DAR of 2 or 4, ensuring that every molecule in the dose is uniform.
- Optimized PK/PD: Uniform molecules result in a stable Pharmacokinetic (PK) profile, allowing the drug to circulate longer without being cleared prematurely by the liver or kidneys.
2. ConjuAll™ Linker: The Ultimate Safety Gate
The linker is the most critical component in preventing “off-target toxicity.” If a linker is unstable, the toxic payload is released into the bloodstream before reaching the tumor, damaging healthy organs.
- Enzymatic Cleavability: LCB’s linkers are designed to be cleaved exclusively by $\beta$-glucuronidase, an enzyme that is highly overexpressed inside lysosomal compartments of cancer cells.
- Plasma Stability: In the neutral pH of human blood, the linker remains 100% intact. This drastically reduces the common side effects of ADCs, such as neutropenia and thrombocytopenia, which are typically caused by premature drug release.
- Widening the Therapeutic Index (TI): By minimizing systemic exposure, LCB can safely administer higher doses, significantly increasing the chances of tumor eradication.
3. Advanced Payload Toolbox: Overcoming Resistance
A delivery system is only as good as the “warhead” it carries. LCB has developed a diverse library of payloads that move beyond traditional microtubule inhibitors (like MMAE).
- Topoisomerase I Inhibitors: LCB has engineered its own Topo-I inhibitors that are highly potent but designed with a “Bystander Effect.” Once the primary cancer cell is killed, the payload can diffuse to neighboring cancer cells that might not express the target antigen, ensuring a more thorough anti-tumor response.
- PBD Prodrugs: For hard-to-treat, refractory cancers, LCB utilizes Pyrrolobenzodiazepine (PBD) dimers. Unlike competitors’ PBDs, LCB’s PBD Prodrug is inactivated until it enters the cancer cell, further enhancing the safety margin.
4. Strategic Engineering: The “Plug-and-Play” Scalability
What makes LigaChem’s platform truly powerful is its modularity.
- Antibody Agnostic: The ConjuAll™ chemistry can be applied to almost any antibody—whether it is an IgG1, IgG4, or even a bispecific scaffold.
- Collaborative Synergy: This versatility is why global leaders like Janssen (targeting Trop2) and Amgen have chosen LCB’s platform. It allows them to take their existing proprietary antibodies and transform them into best-in-class ADCs with minimal re-engineering.
Technical Summary: The “Best-in-Class” Edge of LigaChem Bio
In the 2026 ADC landscape, the market has moved beyond early-stage concepts. Top-tier status is now defined by Therapeutic Index (TI) expansion and the ability to overcome resistance to first-generation blockbusters like Enhertu. LigaChem Bio stands at the pinnacle of this shift.
1. Platform Comparison: LCB vs. Global Leaders
While current market leaders like Daiichi Sankyo have revolutionized the field with high-DAR (DAR 8) technologies, LCB provides a superior alternative for next-generation precision.
| Feature | Current Top-Tier Trend (e.g., DXd Tech) | LigaChem Bio (ConjuAll™) | The Top-Tier Advantage |
| Conjugation Strategy | Often stochastic or semi-site-specific | True Enzyme-Mediated Site-Specific | Higher Homogeneity: LCB produces a pure single-DAR (2 or 4) species, ensuring consistent drug behavior and easier manufacturing scaling. |
| Linker Stability | Peptide-based (Protease cleavable) | beta-Glucuronidase Cleavable | Superior Plasma Stability: LCB’s linker is more stable in systemic circulation, drastically reducing “off-target” toxicity (like interstitial lung disease) common in current ADCs. |
| Payload Potency | High-DAR / Lower Potency strategy | Low-DAR / High-Potency strategy | Widened Therapeutic Index: By using more potent drugs at a lower DAR, LCB achieves the same efficacy with less total drug exposure, improving patient safety. |
2. Strategic Differentiation: Overcoming “Enhertu-Resistance”
One of the most significant technical moats for LCB in 2026 is its performance in refractory patients.
- Clinical Superiority: LCB’s HER2 ADC (IKS014/LCB14) recently made headlines by showing a 75% Objective Response Rate (ORR) in patients who had already failed or become resistant to Enhertu (Trastuzumab Deruxtecan).
- The “Bystander” Evolution: LCB’s payloads feature a sophisticated Bystander Effect—once the drug kills the target cell, it can cross into neighboring cells to kill “antigen-low” tumors, effectively tackling the problem of tumor heterogeneity that plagues first-generation ADCs.
3. Scalability & Modality Expansion
LCB’s platform is designed as a “Plug-and-Play” system, which is why it has won the “Best ADC Platform Technology” award at the World ADC Summit for seven consecutive years.
Third-Party Validation: The fact that Janssen (a leader in oncology) paid over $1.7B for LCB84 underscores that the industry views LCB’s technology not as an “alternative,” but as a mandatory upgrade for future oncology pipelines.
Modularity: It can be seamlessly integrated with bispecific antibodies (BsADCs) or even STING agonists (Immunostimulatory ADCs).


Reference: https://www.legochembio.com/rnd/technology_01.php?lang=e
Corporate Presentations
LigaChem Biosciences_Corporate Presentation_4Q Eng_Official
Reference: https://ligachembio.com/invest/irdata.php?lang=e&cate=1
Valuation Report: LigaChem Bio (2026.01.28)
The Global Standard of ADC: Transitioning from Platform Licensor to Clinical Powerhouse
As of January 28, 2026, LigaChem Bio (LCB) is being re-rated as a “Global Tier-1” oncology player. With the successful initiation of the Phase 2 trial for LCB84 (Trop2-ADC) by Janssen and the high-DAR pipeline expansion, LCB is moving beyond a “tech-provider” to a company that commands recurring royalty-based enterprise value.
1. rNPV Analysis: Sum-of-the-Parts (SOTP) Valuation
The rNPV (risk-adjusted Net Present Value) for LigaChem Bio reflects its highly diversified risk profile, with over 15 active out-licensed programs.
| Asset / Platform | Target | Stage | Est. POS | rNPV Valuation |
| LCB84 (Janssen) | Trop2 ADC | Phase 2 | 28% | ~$2.1B (KRW 2.8T) |
| LCB14 (Fosun/Iksuda) | HER2 ADC | Phase 3 (CN) | 75% | ~$1.3B (KRW 1.8T) |
| LCB71 (CStone) | ROR1 ADC | Phase 1/2 | 20% | ~$1.0B (KRW 1.4T) |
| ConjuAll™ Platform | ADC Infrastructure | Multiple Deals | 45% | ~$2.6B (KRW 3.5T) |
| Pre-clinical/Early Assets | TPD / ISAC | Early Stage | 5% | ~$0.7B (KRW 1.0T) |
| Total Pipeline Value | ~$7.7B (KRW 10.5T) |
- Net Cash Position: ~$0.6B (KRW 0.8T), following a series of milestone payments in Q4 2025.
- Implied Enterprise Value (EV): Approximately $8.3B (KRW 11.3T).
- Estimated Fair Market Cap: KRW 12T ~ 14T range, depending on Phase 2 data readouts.
2. Technology-Driven EBITDA & Multiples
LigaChem Bio has established a “Milestone Machine” business model, generating consistent cash flow that is rare in the biotech sector.
- 2026 Est. Revenue: ~$380M (KRW 510B) — Driven by a $100M milestone from Janssen and new upfronts from the recently signed Ono Pharmaceutical deal.
- 2026 Est. EBITDA: ~$210M (KRW 280B) — Significant operating leverage as global partners fund the expensive late-stage clinical trials.
- Target Multiple: Top-tier ADC peers (e.g., Genmab, Seagen historicals) trade at 40x – 45x due to the high barrier of entry in ADC chemistry.$210M (EBITDA) × 40 = ~$8.4B (KRW 11.4T)
3. Why the “Premium” is Justified: The Best-in-Class Moat
- Chemical Superiority (ConjuAll™):The market rewards LCB’s Site-Specific Conjugation. By ensuring a fixed DAR (Drug-to-Antibody Ratio) of 2 or 4, LCB provides a level of safety and manufacturing consistency that “stochastic” (random) ADC competitors cannot match.
- Safety as a Competitive Edge: While competitors struggle with “off-target” toxicities (like lung or liver damage), LCB’s beta-glucuronidase-cleavable linker ensures the toxic payload is only released inside the tumor. This allows for a much wider Therapeutic Index (TI), enabling higher and more effective dosing.
- Validation by the “Big Four”: With Janssen (J&J), Amgen, GSK, and Ono Pharmaceutical all utilizing LCB’s linkers, the platform has become the “Industry Standard.” In 2026, a “LigaChem Inside” label on a drug candidate significantly lowers the perceived risk for institutional investors.
4. Strategic Conclusion
Market Snapshot [2026.01.28]:
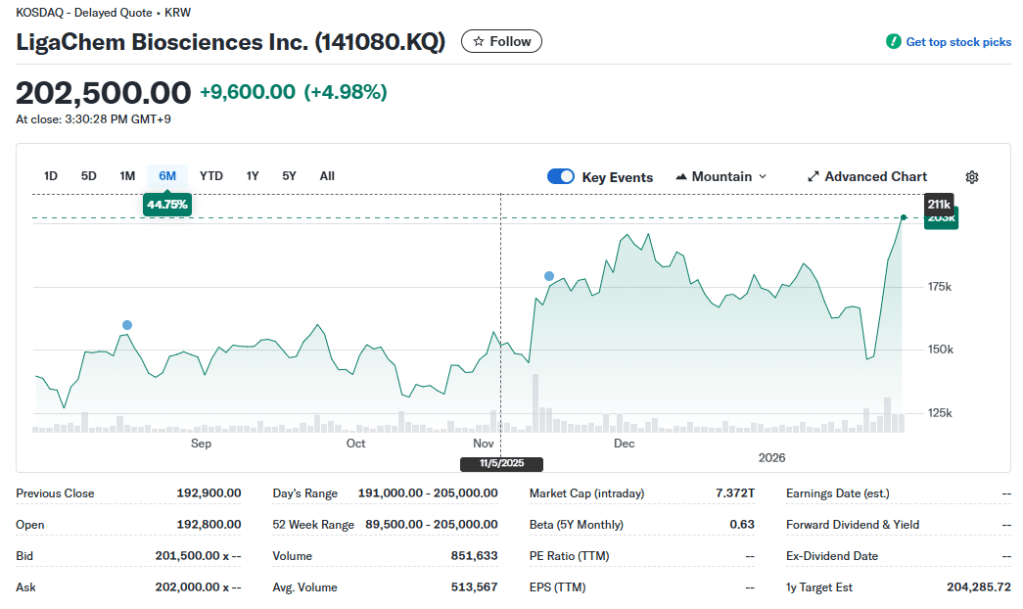
- Current Status: Consolidation near the KRW 10T mark.
- Investment Verdict: Strong Buy on Clinical De-risking.
LigaChem Bio is currently priced as a “Successful Platform Company.” However, the “Alpha Growth” will come from the first clinical data of LCB84 (Trop2) expected in mid-2026. If the data shows superiority over competitors like Dato-DXd, LigaChem Bio will likely leapfrog to a KRW 20T+ valuation, following the trajectory of global ADC pioneers.

댓글 남기기