1. LAPSCOVERY™: The Verified Bio-Platform
Hanmi’s core technical advantage is the LAPSCOVERY™ (Long-Acting Protein/Peptide Discovery) technology.
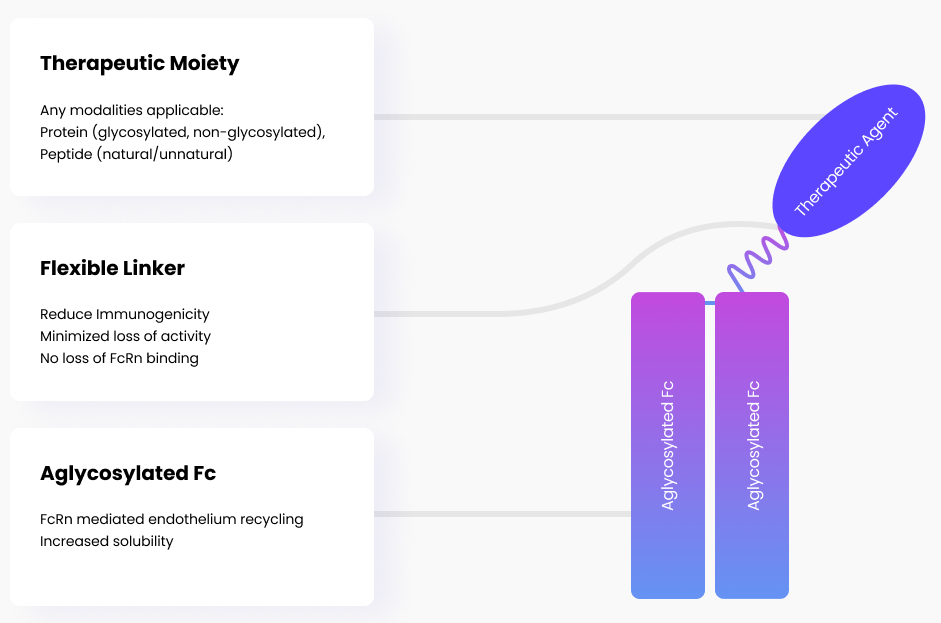
- Mechanism: It utilizes a non-glycosylated IgG4 Fc fragment as a “shuttle” to extend the half-life of biologics.
- Technical Maturity: Unlike many biotech platforms still in the “concept” phase, LAPSCOVERY is fully commercialized and FDA-verified (via Rolvedon/Rolontis). This verification significantly lowers the “technical risk” for its next-generation metabolic assets.
- 2026 Status: The platform is now scaling into the Triple Agonist era, targeting MASH and obesity simultaneously with a single molecule.
LAPSCOVERYTM
Long Acting Protein / Peptide Discovery Platform Technology
Overview
Biologics have a short half-life when administered to the human body. This results in the need for frequent injections and inconvenience to patients. LAPSCOVERYTM (Long Acting Protein/Peptide Discovery) is a platform technology that prolongs the active duration of biologics and makes it possible for once-weekly or monthly drug administration.
Reference: https://www.hanmipharm.com/science/manufacturing/lapscoverytm.hm
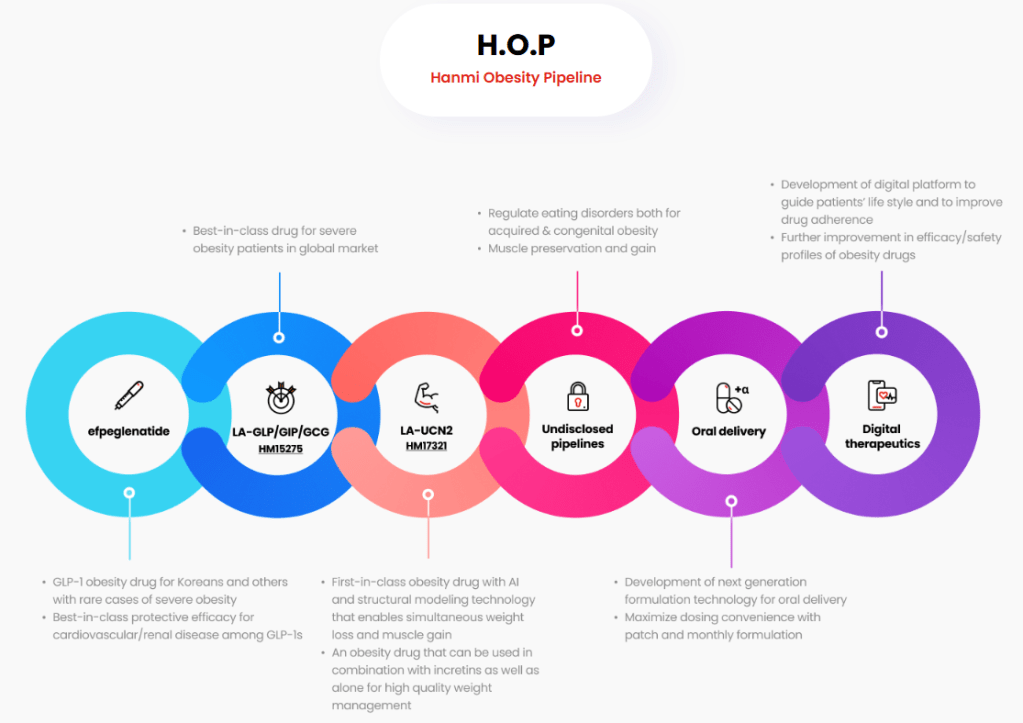
2. H.O.P (Hanmi Obesity Pipeline): The Self-Sustaining Ecosystem
Hanmi’s H.O.P Project is a technically holistic approach to obesity that spans from weight loss to muscle preservation.
- Efpeglenatide (LAPS-Exendin-4): Targeted for H2 2026 Korean launch.
- The “Tolerability” Edge: Clinical trials have shown that Efpeglenatide offers a superior GI (Gastrointestinal) safety profile compared to competitors like Wegovy. This is technically significant because “treatment adherence” is the biggest barrier in long-term obesity management.
- Manufacturing Superiority: Produced at the Pyeongtaek Bio Plant, which features a massive 12,500L microbial culture system. This ensures a stable supply and high-margin production, avoiding the “supply bottleneck” issues seen by global peers.
- Next-Gen HM15275 (Triple Agonist): Benchmark against Eli Lilly’s Retatrutide. HM15275 aims for >20% weight loss while minimizing metabolic rebound through glucagon-mediated energy expenditure.
Reference: https://www.hanmipharm.com/science/reaserach-area.hm
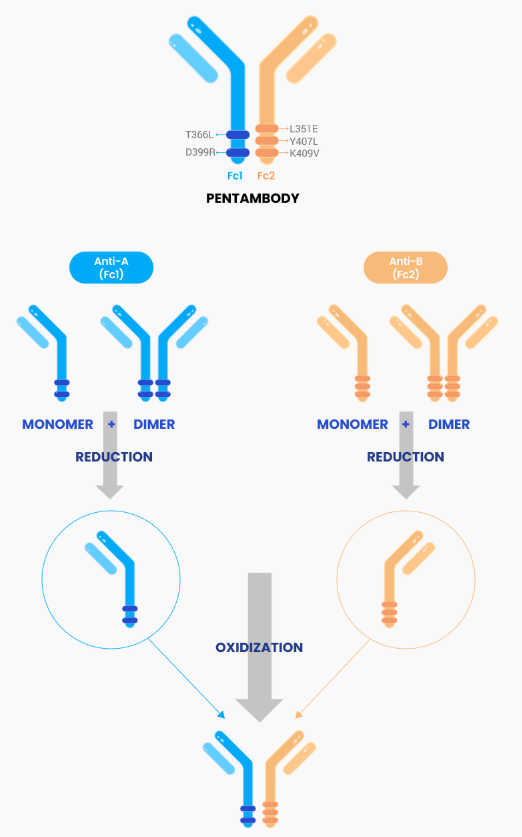
3. PENTAMBODY™: Entering the Bispecific Oncology Arena
- BH3120 (PD-L1 x 4-1BB): As of Jan 2026, the global Phase 1 trial co-developed with Beijing Hanmi has confirmed early clinical efficacy and a robust safety profile.
- Technical Breakthough: BH3120 successfully “decouples” tumor-specific immune activation from systemic toxicity (a common failure point for 4-1BB targets), proving the precision of the PENTAMBODY™ structural design.
Bispecific antibody platform technology
Penta amino acid mutated bispecific antibody
Overview
PENTAMBODY is a next generation, bispecific antibody platform technology developed by Beijing Hanmi Pharmaceutical Co., a subsidiary of Hanmi. This platform allows the antibody to simultaneously bind two different targets for both immuno-oncology therapy and targeted therapy. It possesses natural IgG-like antibody structure, therefore demonstrating good stability and other important natural IgG-like biophysical properties. Multiple bispecific antibodies using PENTAMBODY are being developed by Hanmi.
Reference: https://www.hanmipharm.com/science/manufacturing/pentambodytm.hm
Valuation Report: Hanmi Pharmaceutical (2026.02.03)
Investment Rating: Accumulate (Transitioning to a “Tech-Growth” Multiple)
The market currently values Hanmi primarily as a “Traditional Pharma” with high-performing domestic sales. However, our rNPV analysis incorporates the “Platform Premium” of H.O.P and MASH.
1. Adjusted rNPV Analysis (Sum-of-the-Parts)
Calculated at 1 USD = 1,480.80 KRW.
| Asset Segment | Core Technology | Strategic Value | rNPV (USD) | rNPV (KRW) |
| Operating Value | Rosuzet, Amosartan, Beijing Hanmi | Cash Cow Pillar | ~$3.11B | ~KRW 4.60T |
| H.O.P (Obesity) | Efpeglenatide (Launch 2H26) | Market Leader | ~$1.22B | ~KRW 1.80T |
| MASH Pipeline | Efinopegdutide (MSD Partnered) | Ph 2b Readout 2026 | ~$0.64B | ~KRW 0.95T |
| Oncology/Rare | BH3120 & Triple Agonists | Early Clinicals | ~$0.17B | ~KRW 0.25T |
| Total Enterprise Value | ~$5.14B | ~KRW 7.60T |
2. Strategic Valuation Drivers for 2026
- Domestic Launch (Efpeglenatide): Revenue of KRW 100B+ within 12 months of launch expected. Unlike imported obesity drugs, Hanmi’s internal manufacturing allows for aggressive pricing and a wider margin.
- MASH Momentum: Efinopegdutide (licensed to MSD) is currently analyzed for Phase 2b data. High liver fat reduction (72.7% in Ph 2a) suggests it could become a top-tier therapy in the $25B MASH market.
- Governance Stability: The transition to professional management has eliminated the “Governance Discount,” allowing R&D investment to be more focused and transparent.
3. Strategic Conclusion
“Platform Value is the True Upside”
Hanmi Pharmaceutical is currently trading at a discount compared to our KRW 7.6T valuation. The market is underestimating the “Manufacturing Moat” of the Pyeongtaek Plant and the “Digital Synergy” of its H.O.P project.
Final Summary:
“2026 is the year Hanmi’s R&D transforms into real-world revenue. With Efpeglenatide’s launch and critical MASH data readouts, the current price represents a high-probability entry point. We maintain an Accumulate rating as the company transitions from a ‘Legacy Pharma’ to a ‘Global Tech Powerhouse’.”
IR Presentation
2025 Hanmi Vision Day
Reference: https://www.hanmipharm.com/about/investor-relations/ir/event/detail-341.hm

댓글 남기기